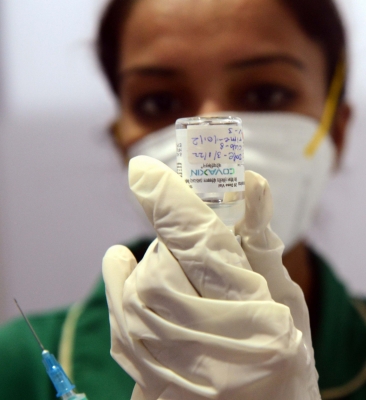
New Delhi, April 26 : The Drugs Controller General of India (DCGI) on Tuesday approved the restricted emergency use authorisation (EUA) to Bharat Biotech’s Covaxin for children between the age of 6-12 years, sources said.
The DGCI has asked vaccine manufacturer to submit safety data including the data on adverse event with due analysis every 15 days for the first two months and monthly thereafter upto 5 months, said the source familiar with the development.
This development comes after the Subject Expert Committee (SEC) discussed recommendations for the restricted emergency use of Covaxin in children aged between 2-12 years.
However, the SEC, after a meeting on Thursday, had recommended restricted emergency use of Biological E’s Covid-19 vaccine, Corbevax, in children aged 5-12 years.
avr/svn/
#DCGI #Covaxin #Delhi #Delhi #New Delhi #Covid-19